|
Therefore, the electronic configuration of Z n2+ is 1s 22s 22p 63s 23p 63d 10. have been investigated theoretically in more detail. dipole: Any object (such as a magnet Adding Custom. The band structures of Zn-based hydroxyapatites co-doped with Mg at varying amounts from 0 to 2.0 at. In this case, all the 4p subshells are empty hence, we start by removing from the s orbital, which is the 4s orbital. 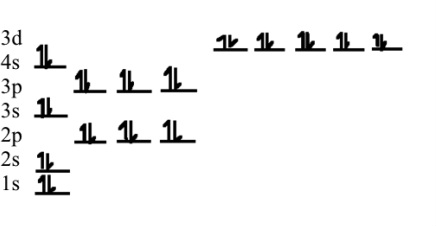
Since we need to take away two electrons, we first remove electrons from the outermost shell (n4). Hence, the electron configuration for Ca 2+ is 1s 2 2s 2 2p 6 3s 2 3p 6. Zn 2+ electronic configuration – 1s 22s 22p 63s 23p 63d 10 Because the number of protons remains unchanged when an atom forms an ion, the atomic number of the element must be 13. ion is, Explanation : Formula used : where, V number of valence electrons present in central atom. The calcium ion (Ca 2+), however, has two electrons less. For the d-block elements, the outermost s-sublevel has. Zinc is a d-block element, also known as a transition element. The electron configuration of Zn2+ is 1s22s22p63s23p63d10. The Zn2+ ion has lost two electrons, which leaves it with 30 protons and 28 electrons. We have to remove two electrons from zinc neutral atom to get electronic configuration of The electron configuration of a neutral zinc atom is 1s22s22p63s23p63d104s2. Zn electronic configuration – 1s 22s 22p 63s 23p 64s 23d 10 If an ion has a negative charge, we have to add the electrons from the neutral electronic configuration of the element.Ģ+ charge indicates substraction of two electrons from neutral electronic configuration. 
If an ion has a positive charge, we have to subtract the electrons from the neutral electronic configuration of the element. number of valence electrons that the element iodine has 2) How many iodide ions are needed to cancel out the charge on a magnesium ion 2g) How is the. We can determine the ion’s electron configuration by first figuring out the neutral atom’s electron configuration, then adding or deleting electrons based on the ion’s charge. To write the configuration for the Zinc and the Zinc ion, first we need to write the electron configuration for just Zinc (Zn). Ground state electronic configuration(Neutral atom)- 1s 22s 22p 63s 23p 64s 23d 10 IonĪn ion is an electrically charged particle formed by withdrawing or adding electrons from a neutral atom or adding electrons to a neutral atom to make a negative ion. What is the Electron Configuration for ZN 2+? The given element is Zinc.Įlectronic configuration – 1s 22s 22p 63s 23p 64s 23d 10 or 3d 104s 2
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
